Introduction
Materials and Methods
Animals, experimental design, management, and diet
Sample collection and analyses
Sows reproductive performance
Statistical analyses
Results
Sow performance
Litter performance
Milk composition
Discussion
Conclusion
Introduction
The genetic progress of highly prolific sows has created a new nutritional paradigm for the swine industry. The severe mobilization of maternal body reserves is a consequence of the increased lactational demands placed on modern sows by their greater milk potential and larger litters (Rutherford et al., 2013; Oh et al., 2021). Litter development predominantly occurs during late gestation and is linked to a significant increase in essential amino acids (eAA) requirement (Guan et al., 2004; Le Dividich et al., 2005). This period of immense metabolic demand is critical, it directly governs piglet growth, pre-weaning survival, and the sow’s ability to re-breed successfully (Theil et al., 2012). Previously, with lower litter size (LLS), the gestation requirement could be met with a constant dietary allowance. However, with high litter sizes (HLS), this approach is no longer sufficient, and it is now necessary to adjust the daily nutrient supply. This can be achieved through a modification of feed quantity or composition of the diet (NRC, 2012; Hurley, 2015; Schmitt et al., 2019).
Effective precision feeding must account for the dynamic nature of nutritional requirements throughout each physiological stage from one sow to another (Wellington et al., 2023). The eAA were originally considered as substrates for protein synthesis and retention, however, recently attention has been given to their additional role as intermediates in key molecules that influence the efficiency of organs critical development. They also support milk protein synthesis and metabolic precursors for key bioactive compounds that regulate lactation and recovery (Kim et al., 2013; Strathe et al., 2020). Among these, L-phenylalanine (Phe) is of particular interest because it could act as a direct metabolic precursor for tyrosine (Tyr), an irreversible step catalyzed by phenylalanine hydroxylase (PAH) (Horne et al., 2002). Which in turn is critical for the synthesis of key catecholamines (e.g., adrenaline, noradrenaline, and dopamine) and the thyroid hormone thyroxine (T4) (Liu et al., 2018; Luise et al., 2023). Phe affects T4 indirectly via PAH by acting as a precursor for Tyr (Shebl et al., 2024). Animal studies show that Phe and Tyr deficiency can reduce T4 levels when growth is severely impaired (Elkin, 1981). In dairy cows, omitting Phe from an eAA mixture significantly reduces milk protein yield compared to complete eAA supplementation (Doepel et al., 2016; Kim et al., 2020; Fazio et al., 2022). While previous work by Lellis and Speer (1987) suggest that supplementing Phe above requirements does not further increase T4 production in sows, this finding was established under a lower lactational demand than those faced by modern hyper-prolific sows (Gauthier et al., 2019). Phe potential is relevant for hyper-prolific sows bearing large litters, where metabolic and endocrine stress is most acute. The magnitude of lactational stress is linked to litter size (LS), with sows nursing more piglets facing greater nutrient drain and metabolic challenges that often leave weaker piglets with insufficient colostrum intake (Schmitt et al., 2019). High-yielding sows may prioritize eAA for milk protein synthesis over their own body maintenance, and this would further compromise piglet growth.
In this study, we hypothesized that Phe supplementation may be contingent upon LS burden. Our objective was to determine the effects of two supplemental levels of Phe, added to a conventional corn-soybean meal basal diet, on sow body weight (BW) and backfat thickness (BF) loss, litter growth performance, milk macronutrient composition, and T4. We aimed to evaluate whether the response to dietary Phe was modulated by the scale of lactational demand.
Materials and Methods
The experimental procedures were sanctioned by the institutional animal care and use committee of Kangwon National University, Chuncheon, republic of Korea (protocol no. KW-240715-4) and conducted in strict adherence to the South Koreas regulations for the administration of affairs pertaining to experimental animals.
Animals, experimental design, management, and diet
This study was conducted on a commercial pig farm in Haman, South Korea. Thirty-six multiparous Duroc × Landrace × Yorkshire sows (BW 225.22 ± 17.77 kg; parity 3; gestation length: 114 - 116 days) were stratified pre-farrowing into two groups based on expected LS from farm records (LLS group: 11 - 15 piglets per sow, n = 18; and a HLS group: 16 - 20 piglets per sow, n = 18). This classification, based on farms normal distribution and prolific sow literature, ensured balanced representation and allowed evaluation of Phe effects across different reproductive potentials. Sows were housed individually in stalls (2.05 × 1.08 m) in a completely randomized block design, with LLS and HLS serving as the blocking factor. Within each block, sows were randomly assigned to one of three dietary treatments (n = 6 sows per treatment per block). The dietary treatments were: (1) corn-soybean meal basal diet (CN) formulated to meet NRC (2012) guidelines; (2) CN + 0.2% Phe (CN2); (3) CN + 0.4% Phe (CN4). Sows received experimental diets from gestation day 90 until the 21st lactation. The calculated and determined nutrient basal diet composition are provided in Table 1. At gestation day 90, sows were relocated to climate-controlled farrowing rooms and housed individually (2.05 × 1.08 m) in crates that were equipped with a slatted plastic floor. Environmental conditions included a 14-h light and 10-h dark cycle. From day 90, 1.5 kg of feed was provided (single-space feeder fitted with nipple drinkers delivering 1.5 - 2 L·min-1) at 06:00 and 16:00 h, which was gradually increased by +0.4 kg·d-1 until farrowing. A supplemental infrared lamp was installed in each crate to ensure thermal comfort for piglets.
Table 1.
Ingredient and calculated chemical composition of lactation sow diets (as-fed basis).
| Item | Basal diet |
| Ingredients (%) | |
| Corn | 57.87 |
| Wheat bran | 5.00 |
| Soybean meal | 27.88 |
| Animal fat | 3.57 |
| Salt | 0.50 |
| Sugar | 3.00 |
| Limestone | 0.96 |
| Dicalcium phosphate | 0.79 |
| Choline chloride | 0.05 |
| Vitamin premixy | 0.11 |
| Mineral premixz | 0.22 |
| Phytase | 0.05 |
| Total | 100.00 |
| Calculated chemical composition (%) | |
| ME (kcal·kg-1) | 3,300 |
| CP | 18.00 |
| Crude fat | 5.80 |
| Ash | 3.96 |
| Ca | 0.76 |
| P | 0.65 |
| Lys | 0.96 |
| Met | 0.28 |
| Phe | 0.91 |
Sample collection and analyses
Blood samples were collected via jugular venipuncture from three sows per dietary group on days 0, 14, and 21 of lactation using heparin-free 10-mL vacutainer tubes (Becton Dickinson, USA). Serum was separated by centrifugation (3,000 rpm, 10 min, 37℃; Heraeus Biofuge 22R, Heraeus Sepatech GmbH, Germany) between 09:00 - 10:00 h and stored at -20℃. Concentrations of total T4 in serum were determined using analyte specific commercial radioimmunoassay (RIAs; MP Biomedical, USA), as prescribed in porcine samples (Pasternak et al., 2020). Milk samples (20 mL/sow) were collected from three randomly selected sows per group at days 0, 3, and 10 (within 10 h postpartum), of lactation. Samples were obtained from all teats on one mammary side 10 min post-injection of 2 mL oxytocin (10 IU·mL-1, intramuscular). Pooled samples were homogenized, stored at -20℃, and analyzed for protein, fat, lactose, and total solids using a MilkoScan FT-120 analyzer (FOSS Electric, Denmark).
Sows reproductive performance
Sow BW and BF were recorded at gestation day 90, within 24 h postpartum, and at weaning. The BF was ultrasonographically measured (Sonolayer SAL-32B, Toshiba, Japan) 65 mm lateral to the midline at the last rib. Piglets were weighed individually at birth and weaning (lactation day 21). Feed refusals were collected daily between 07:00 - 08:00 h, with fresh feed provided immediately. Sow average daily feed intake (ADFI) was determined by subtracting the weight of refused feed from the initial allocation. The weaning-to-estrus interval (WEI) was defined as the interval days between weaning and the onset of subsequent estrus.
Statistical analyses
Data were analyzed using the General Linear Model (GLM) procedure in SAS (2012). The model included the fixed effects of dietary treatment, LS group, and their interaction, with parity as a covariate. Initial sow BW was included as a covariate for analyses of sow performance parameters. The individual sow served as the experimental unit for all analyses. Tukey’s Honestly Significant Difference test was used for post-hoc mean separation when significant main effects (p < 0.05) or a tendency (p < 0.10) were detected. Results are presented as least-squares means ± standard error of the mean.
Results
Sow performance
The results in Table 2 demonstrate that Phe supplementation did not affect sow BW at 24 hours postpartum or at weaning across the CN, CN2, and CN4. The BW loss during lactation was greater for sows with HLS compared to those with LLS (p < 0.05), while Phe treatment had no significant effect. The BF remained consistent among the CN, CN2, and CN4 groups during 24 hours postpartum and weaning. However, the BF loss during lactation showed a higher trend (p = 0.055) in LLS sows compared to LLS sows, with no significant differences observed due to Phe supplementation. The ADFI and weaning to estrus interval were not affected by Phe supplementation or sow LS in the CN, CN2, and CN4 groups.
Table 2.
Effects of phenylalanine supplementation on sow performance during lactating.
Litter performance
Table 3, in LLS and HLS groups, the initial number of piglets born, the number weaned, and preweaning mortality rates did not differ among sows fed the CN, CN2, and CN4 diets. Mortality rates tended to be higher (p = 0.080) in sows with HLS than in those with LLS. Sows with HLS had higher (p < 0.05) initial and weaned piglets. Litter weights at birth and weaning were greater (p < 0.05) in the HLS group, with no effect of Phe supplementation within either group. Piglet weights at birth or during weaning did not differ between LLS and HLS. Furthermore, there was no evidence of an interaction between Phe level and LS group for all measured outcome.
Table 3.
Effects of phenylalanine supplementation on litter performance of lactating sows.
Milk composition
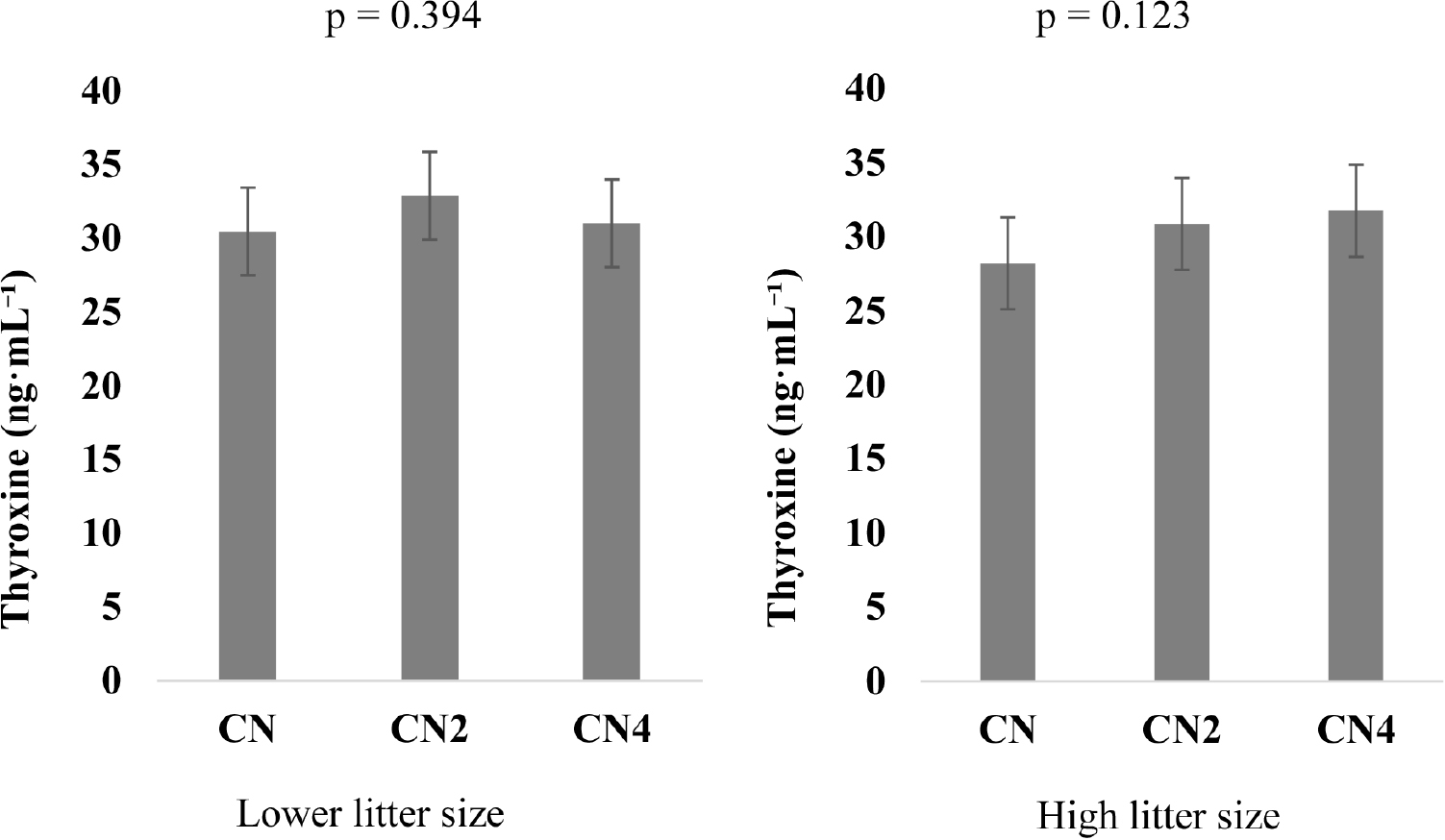
The analysis revealed no differences in the composition of milk fat, protein, lactose, total solids, and solids-not-fat across CN, CN2, and CN4 groups (Table 4). Similarly, in Fig. 1, the T4 levels in sows within LLS and HLS showed no difference in CN, CN2, and CN4.
Table 4.
Effects of phenylalanine supplementation on milk composition of lactating sows.
Discussion
Dietary Phe is an eAA and the primary precursor for Tyr, which plays an important role in proteins synthesis, neurotransmitters production, and hormones regulation in livestock (Ahmed, 2009; Kim et al., 2020; Shebl et al., 2024). In ruminant and growing animals, an improved growth rates have been associated with higher availability of Phe, Tyr, valine, leucine, and isoleucine (Imaz et al., 2022). During lactation, sows have an increased requirement for eAA to support milk synthesis, endocrine function, immune response and overall metabolic activity (Guan et al., 2004; Tokach et al., 2019). However, in this study supplementing sows diets with 0.2% or 0.4% Phe from day 90 gestation through lactation did not improve BW, BF, ADFI, WEI, litter performance, or milk composition. These findings suggest that the basal corn-soybean meal diet was sufficient in meeting the sows Phe requirements during late gestation and lactation (NRC, 2012). When compared with previous studies, the supplementation levels used in this experiment were moderate relative to the total dietary Phe supply and exceeded NRC (2012) requirement estimates but remained within physiologically reasonable ranges. Unlike studies in which amino acid imbalances or deficiencies were deliberately induced (Elkin, 1980; Kim et al., 2013), the present diets were nutritionally adequate, which may explain the limited response. In such contexts, surplus Phe is likely oxidized or redirected toward Tyr synthesis without exerting additional effects on reproductive or lactational performance (Rezaei et al., 2013). Instead, the most effects were attributable to LS, whereby sows nurturing larger litters experienced greater BW and BF loss due to the immense energy demands of milk production (Seoane et al., 2020; Oh et al., 2021; Imaz et al., 2022). This energy deficit overshadowed the marginal benefit that could be derived from Phe supply. The trend toward higher preweaning mortality recorded in large litters further support the concept that energy availability and milk yield, rather than individual amino acid supply, are dominant limiting factor under these condition (Le Dividich et al., 2005; Rutherford et al., 2013).
The lack of treatment effects on piglet growth and survivability is also consistent with the absence of changes in milk yield or composition. Sow milk composition is tightly regulated and remains relatively stable across a wide range of dietary conditions, provided that no severe eAA deficiency exists (Theil et al., 2012; Hurley, 2015). Previous studies demonstrate that mammary gland homeostatic mechanisms buffer against dietary variation, maintaining consistent amino acid profiles and macronutrient concentrations (Daza et al., 2004; Zhou et al., 2018). Thus, supplementation with 0.2% or 0.4% Phe was insufficient to disrupt this regulatory control in sows fed nutritionally balanced diets.
Litter outcomes are multifactorial and involve a complex genetic, nutritional, and environmental interplay (Milligan et al., 2002). During lactation, eAA prioritization could favour milk production and compromise other metabolic processes (Hou et al., 2015; Hussain et al., 2020). Furthermore, the lack of effect on ADFI and WEI could mean that Phe supplementation did not alter the sow’s fundamental energy status, which is the primary driver of postpartum reproductive recovery.
The rise in sows prolificacy has been associated with a decrease in average BW and the rise in within-litter variation of individual birth weight (Quiniou et al., 2002). There was no improvement in the initial number of piglets born and the number weaned in all treatments. This outcome is closely connected to the absence of any measurable change in milk yield and composition resulting from dietary Phe supplementation. Piglet growth during lactation is fundamentally determined by the quantity and quality of sow milk. It is regulated for its macronutrient content, including eAA, fat, protein, and lactose to ensure they remain within optimal physiological ranges regardless of variations in diet unless there is a marked eAA deficiency (Theil et al., 2012; Hurley, 2015). Previous studies confirm that sow milk composition is highly conserved throughout the lactation period and across different feeding regimens. The regulatory mechanisms within the mammary gland buffer against change, maintaining consistent amino acid profiles and ratios (Daza et al., 2004). The findings by Zhou et al. (2018) suggest that the homeostatic control means substantial dietary imbalances are likely to impact milk content. This provides the reasons why supplemental diets with 0.2% or 0.4% Phe did not enhance piglet growth or milk nutrient composition in sows fed balanced diets.
One proposed mechanism by which Phe supplementation might influence milk and piglet outcomes is through its role in Tyr and T4 synthesis (Liu et al., 2018; Jing and Zhang, 2022; Luise et al., 2023). T4 is a thyroid hormone that promotes the stimulation of milk synthesis in animals through two mechanisms, first, direct action on the mammary gland to upregulate milk production, and synergistic action with prolactin to maximize metabolic efficiency (Doepel et al., 2016; Kim et al., 2020; Fazio et al., 2022). Therefore, it was hypothesized that increasing Phe availability would boost T4 production, which will directly affect metabolic and lactational performance. This study did not achieve this outcome in maternal T4 levels, although it was numerically high. A study by Elkin (1980) reported that only severe deficiencies in eAA can depress serum thyroid hormone levels and impair growth. Such extreme nutritional deficits are not characteristic of modern nutritionally balanced sow feed diets, which are specifically formulated to meet the increased metabolic demands of lactation.
Conclusion
In conclusion, supplementing Phe at 0.2% or 0.4% above requirement in a corn-soybean meal diet formulated to meet NRC (2012) recommendations did not improve sow performance, litter growth, or milk composition in multiparous hyperprolific sows. The numerical, increase in maternal T4 concentrations indicates that any endocrine response to Phe at these levels is likely subtle and warrants further research. Given the sample size limitation, these findings suggest that the primary challenge for modern hyper-prolific sows is managing the severe energy deficit associated with nurturing large litters, an issue which Phe supplementation alone could not alleviate.