Introduction
Materials and Methods
Animals and experimental design
The source of monzogranite
Animal management
Nutrient digestibility
Excreta sampling and DNA extraction
16s rRNA amplification and sequencing
Statistical analysis
Results
Growth performance
Nutrient digestibility
Microbiota diversity
Fecal microbiota
Discussion
Conclusion
Introduction
The pig gut environment plays a vital role in maintaining the host immune system, nutrient absorption, and growth performance by supporting a diverse bacterial community (Nowak et al., 2017; Moturi et al., 2021). The weaning period is the most stressful point in pig life and could induce a challenging situation affecting microbes within the gastrointestinal tract (Kim et al., 2022; Hosseindoust et al., 2023). Microbial shifts caused by weaning stress can lead to a rapid proliferation of pathogens in the intestinal tract, resulting in increased inflammation and oxidative responses in the body (Choi et al., 2023; Lee et al., 2024). Furthermore, the undeveloped immune system of young pigs produces high levels of pro-inflammatory cytokines and reactive oxygen species (Shang et al., 2020). A deep and broad understanding of the intestinal structures encompassing microbial synthesis has been achieved by 16S rRNA gene-based analysis (Roh et al., 2010). The next-generation sequencing technology enables us to recognize the diversity of the microbiome and the complex community levels, which allows an explanation of the microbial description in diverse bacterial types (López-Colom et al., 2020).
Monzogranite (MON) is a type of plutonic rock that has similar characteristics to granodiorite containing silica, aluminum tectosilicate, and tectosilicate (Cowie and Walton, 2018). Over the past few years, the effects of MON supplementation in the weaning pig diet have been verified through the physiochemical properties of silicon dioxide in MON (Kong et al., 2004; Liu et al., 2020). Silicon dioxide is known to have high surface activity, greater absorption ability, larger anions, and cation exchange capacity than other minerals in the gut (Wang et al., 2021; An et al., 2023). Furthermore, silicon dioxide is able to bind with toxins, and some pathogens that work as absorbents due to their inner layered structure (Abdel-Wahhab et al., 2002; Abbès et al., 2006). Through these characteristics of silicon dioxide, numerous studies have proven the ability to enhance nutrient utilization and mucosal protection in pigs especially when silica-based mineral including montmorillonite, bentonites, zeolites, and illite is supplemented (Song et al., 2012; Moturi et al., 2021). Notably, the inclusion of silica-based minerals in the swine diet has been shown to increase the population of Lactobacillus while decreasing the population of Escherichia coli (Pieszka et al., 2018; Lee et al., 2020). However, there is a lack of studies about the intestinal environment modulation effects of the different levels of MON on microbiota in weanling pigs. Hence, this experiment was carried out to clarify whether dietary MON supplementation with different levels can change the microbiological structure in weanling pigs and the effects on growth performance.
Materials and Methods
Animals and experimental design
The procedures of this animal experiment were approved by the Institutional Animal Care and Use Committee of Kangwon National University, Chuncheon, South Korea (KW-211022-2).
A total of 160 three-cross-bred [(Landrace × Yorkshire) × Duroc] weanling pigs with 5.40 ± 0.25 kg body weight (BW) were used for this experiment. Weanling pigs were randomly assigned to each treatment with 10 replicates, and 4 pigs were in each replicate. The experimental treatments consisted of a control (basal diet, CON), basal diet + 0.1% MON (MO1), basal diet + 0.2% MON (MO2), and basal diet + 0.3% MON (MO3). Pigs were fed the diet that consisted of corn and soybean meal and was formulated to meet nutrient requirements in NRC (2012) (Table 1). The inclusion level of whey powder was determined based on the previous report by Jeong et al. (2019), who suggested that maintaining dietary lactose levels around 20% is optimal for improving feed intake and growth performance in weaning piglets. This trial was continued for 14 days, and the fecal samples for microbiological analysis were collected through rectal massage at the end of the trial.
Table 1.
Ingredients and chemical composition of basal diet (as-fed).
| Item | Basal diet |
| Ingredient (%) | |
| Corn | 33.14 |
| Lactose | 3.00 |
| Whey powder | 23.08 |
| Soybean meal (45%) | 27.00 |
| Fishmeal (60%) | 3.00 |
| Bakery by product | 5.00 |
| Soy oil | 3.22 |
| Monocalcium phosphate | 0.36 |
| Limestone (fine) | 0.89 |
| Salt | 0.30 |
| L-Lysine·HCl (78%) | 0.27 |
| DL-Methionine (99%) | 0.07 |
| L-Threonine (99%) | 0.01 |
| L-Tryptophan (100%) | 0.11 |
| Choline chloride (50%) | 0.05 |
| Vitamin premixy | 0.25 |
| Mineral premixz | 0.25 |
| Total | 100.00 |
| Calculated composition (%) | |
| Metabolizable energy (kcal·kg-1) | 3,450 |
| Crude protein | 20.00 |
| Lysine | 1.35 |
| Ca | 0.80 |
| P | 0.38 |
y Supplied per kg of diet: 16,000 IU vitamin A (palmitate); 2.00 mg vitamin B1 (thiamin); 5.00 mg vitamin B2 (riboflavin); 2.00 mg vitamin B6 (pyridoxine); 0.03 mg vitamin B12 (cyanocobalamin); 25.00 mg niacin; 0.40 mg folic acid; 0.05 mg biotin; 5.00 mg ethoxyquin; 2,000 IU vitamin D3 (cholecalciferol); 75.00 mg vitamin E (dl-α-tocopheryl acetate); 2.00 mg vitamin K3 (menadione).
The source of monzogranite
The MON was produced by the Korea Institute of Geoscience and Mineral Resources, the MON primarily consists of 66.34% silicon dioxide, 16.44% aluminum oxide, and 3.82% potassium oxide, which was discovered in the area of Mungyeong City, Gyeongsangbuk-do, Republic of Korea.
Animal management
The experimental room was consistently maintained clean. The room temperature was 30℃ at the start of the trial and decreased by 1℃ per week. This trial was carried out for 2 weeks. All of the experimental pigs were weighed individually, and their BW was used for the calculation of average daily gain (ADG). Average daily feed intake (ADFI) was measured by total feed intake and the experimental period.
Nutrient digestibility
To evaluate nutrient digestibility, a Cr2O3 (0.25%) was incorporated into the diets 7 days prior to the conclusion of each experimental phase. Fecal samples were randomly collected from four pigs per pen via rectal stimulation for 3 days prior to the end of each phase. The collected samples were subsequently used to determine the digestibility of dry matter (DM), gross energy (GE), crude protein (CP), and ether extract (EE). Fecal samples were dried in a forced-air oven at 60℃ for 72 h and then ground using a Wiley mill equipped with a 1 mm screen (Thomas Model 4 Wiley Mill, Thomas Scientific, USA). Feed and fecal samples were analyzed in quadruplicate following the standard methods described by AOAC (2005): DM (method 930.15), CP (method 990.03), and EE (method 960.39). The GE content was measured by using bomb calorimeter (Model 1241, Parr Instrument Co., USA).
Excreta sampling and DNA extraction
On day 14 of this experiment, excreta samples of the pig in each replicate were collected through the rectal massage and put in the sterilized 15 mL tubes. All samples were preserved at -80℃ in a deep freezer until the analysis. For the DNA extraction, all the equipment was thoroughly cleaned with 70% alcohol to prepare a sterilized environment, and then 300 mg of each sample was put in the 50 mL tube (Choi et al., 2023). The DNA extraction procedure is conducted with the manufacturer’s instructions in QIAamp PowerFecal Pro DNA Kit (Qiagen, Germany). After the genomic DNA samples were extracted and kept at -80℃ until the following procedure.
16s rRNA amplification and sequencing
The ribosomal RNA (rRNA) amplification in the V1-2 region was performed using Takara Ex-Taq DNA polymerase (Takara Bio, Japan), and the primers 8F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 338R (5′-TGCTGCCTCCCGTAGGAGT-3′). The amplification process was as follows: 33 cycles at 95℃ for 60 s, at 60℃ for 60 s, and at 72℃ for 60 s, followed by a final 10-min cycle at 72℃. The end products, amplicons for PCR products, were separated using the agarose gel electrophoresis (Mupid-One) and purified using the LaboPass™ PCR Purification Kit (Cosmogenetech, Korea). Sequencing of the 16S rRNA gene was performed by using the Ion Personal Genome Machine (Ion PGM). For the quality trimming and de-multiplexing Single-end sequence reads were produced from this sequencing process and then were subjected to quality trimming and de-multiplexing with the utilizing of custom Perl scripts. The processed and quality-trimmed reads were further analyzed using the Quantitative Insights Into Microbial Ecology (QIIME 1.9.1) software. This software facilitated the determination of the richness and diversity indices of the microbial community. Reads matching the Greengenes 13_8 database with 97% sequence identity were classified as operational taxonomic units (OTUs). These OTUs were normalized to 1,033 reads per sample using a single rarefaction. Further visualization and analysis were performed using principal coordinate analysis (PCoA) based on UniFrac distances with EMPeror software. Linear discriminant analysis (LDA) was employed to determine the difference between the abundance of microbial components in two treatments.
Statistical analysis
Data from our study were analyzed statistically using the General Linear Model (GLM) procedure in SAS (SAS, 2013) within a randomized complete block design. Tukey’s honestly significant difference test was employed to differentiate treatment means when significant differences were detected. The pen was considered the experimental unit for analyzing growth performance, while individual piglets were used as the experimental units for the analysis of microbiota. Probability values of p < 0.05 were deemed significant.
Results
Growth performance
The pigs fed different levels of MON did not show any significant difference in ADG, ADFI, and feed efficiency (G/F) (Table 2).
Table 2.
The effects of different levels of monzogranite supplementation on growth performance in weanling pigs.
Nutrient digestibility
No significant differences were observed in the apparent digestibility of DM, GE, CP, or EE among pigs fed diets supplemented with different levels of MON (Table 3).
Table 3.
The effects of different levels of monzogranite supplementation on nutrient digestibility in weanling pigs.
Microbiota diversity
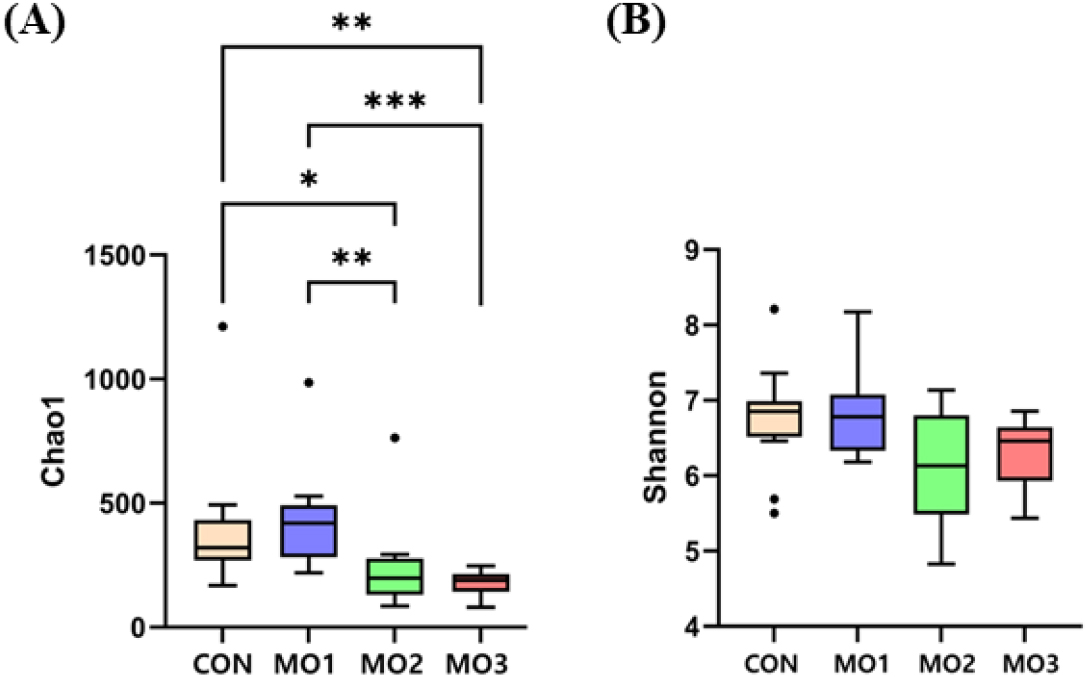
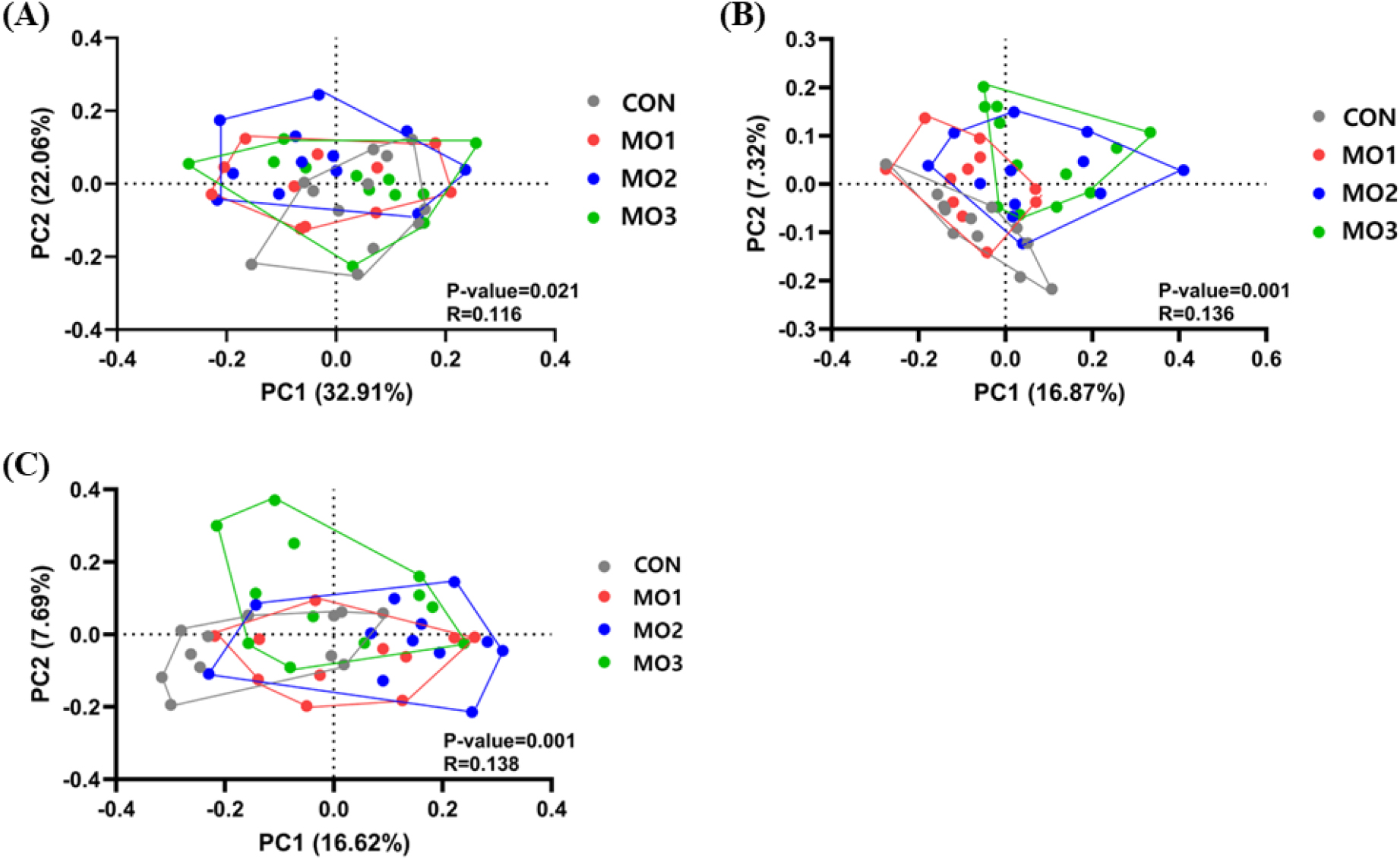
The microbiota alpha diversity of weanling pigs fed the MO2 and MO3 diet revealed a lower Chao 1 index compared to CON and MO1 (p < 0.05; Fig. 1A). On the other hand, the alpha diversity did not show any significant difference in the Shannon-Wiener index (Fig. 1B). The beta diversity of weighted UniFrac distance analysis indicated no significant difference in the pigs fed different levels of dietary MON (Fig. 2A). Unweighted UniFrac distance analysis showed significant differences between CON and MO2, CON and MO3 (Fig. 2B). Bray-Curtis dissimilarity analysis showed significant differences between CON and MO2, CON and MO3 (Fig. 2C).
Fecal microbiota
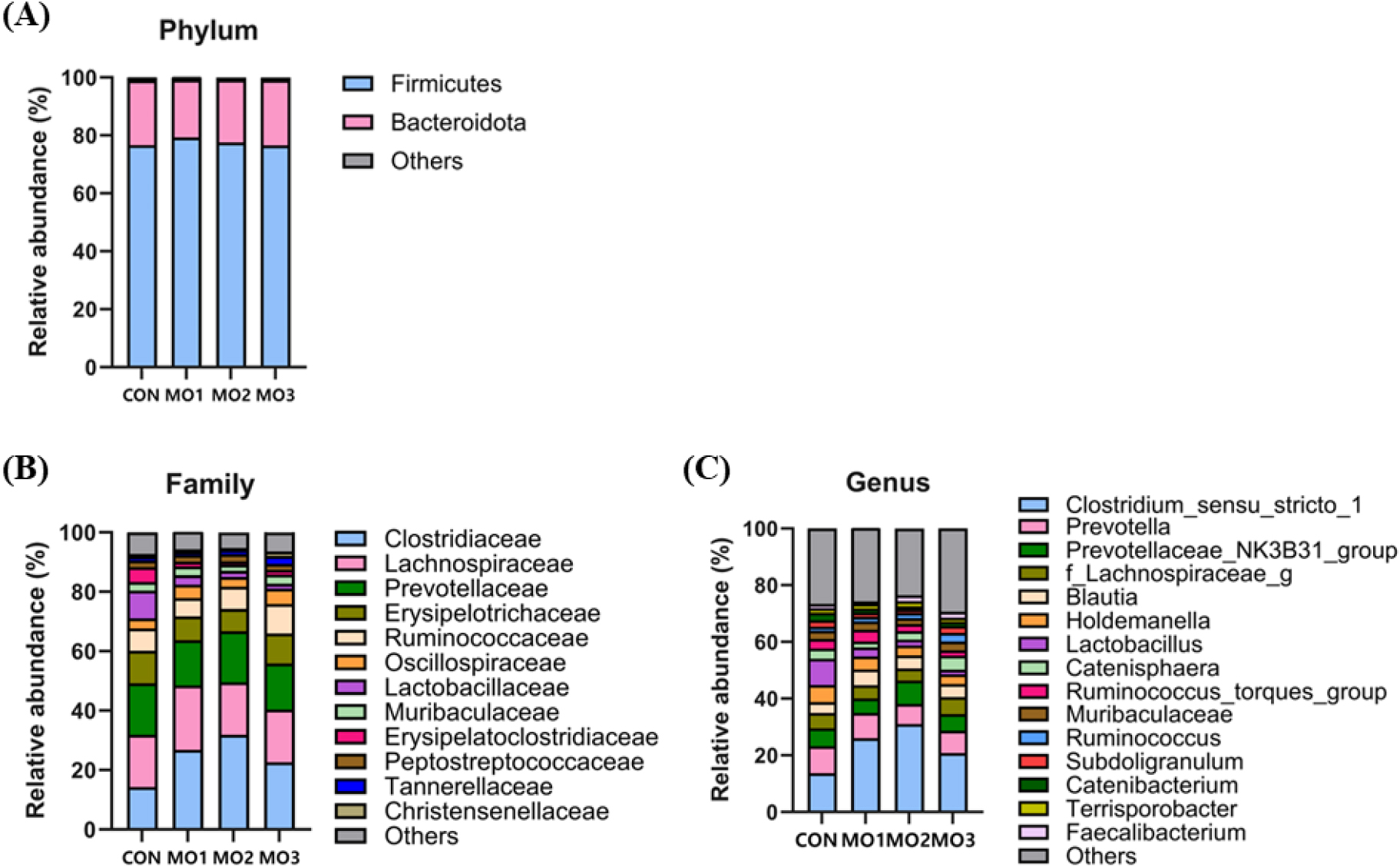
Across the samples with different levels of MON, the relative abundance of Firmicutes was composed of 76.64, 79.29, 77.26, and 76.57% in CON, MO1, MO2, and MO3, respectively (Fig. 3A). Bacteroidota was composed of 22.35, 19.94, 21.63, and 22.56% in CON, MO1, MO2, and MO3, respectively. The relative abundance of Clostridiaceae in MO2 (31.91%) was greater than CON (14.28%), MO1 (26.79%), and MO2 (22.74%) at the family level (Fig. 3B). Moreover, the Lactobacillaceae relative abundance was lower in MO1 (3.16%), MO2 (2.08%), and MO3 (1.63%) than in CON (9.32%) at the family level. At the genus level, the relative abundance of Clostridium_sensu_stricto_1 was significantly greater in MO2 (31.05%) than in CON (13.74%), MO1 (26.03%), and MO3 (20.78%) (Fig. 3C). Furthermore, the relative abundance of Lactobacillus was decreased in MO1 (3.16%), MO2 (2.08%), and MO3 (1.63%) than in CON (9.32%) at the genus level.
In the cladogram of LEfSe analysis of bacterial differences, two groups were compared between CON and MO1 (Fig. 4A), CON and MO2 (Fig. 4B), and CON and MO3 (Fig. 4C). 5 and 1 taxa were identified in CON and MO1, respectively. 10 and 5 taxa were identified in CON and MO2, respectively. 9 and 3 taxa were identified in CON and MO3, respectively. To distinguish taxonomic differences between the different levels of MON supplementation, 11 and 4 phylotypes in the CON and MO1 (Fig. 4D), 20 and 7 phylotypes in the CON and MO2 (Fig. 4E), and 15 and 9 phylotypes in the CON and MO3 (Fig. 4F) were shown as LDA scores. The MO2 group was enriched with the Clostridiales and Clostridia, whereas the CON group was enriched with the Erysipelotrichi and RF39.
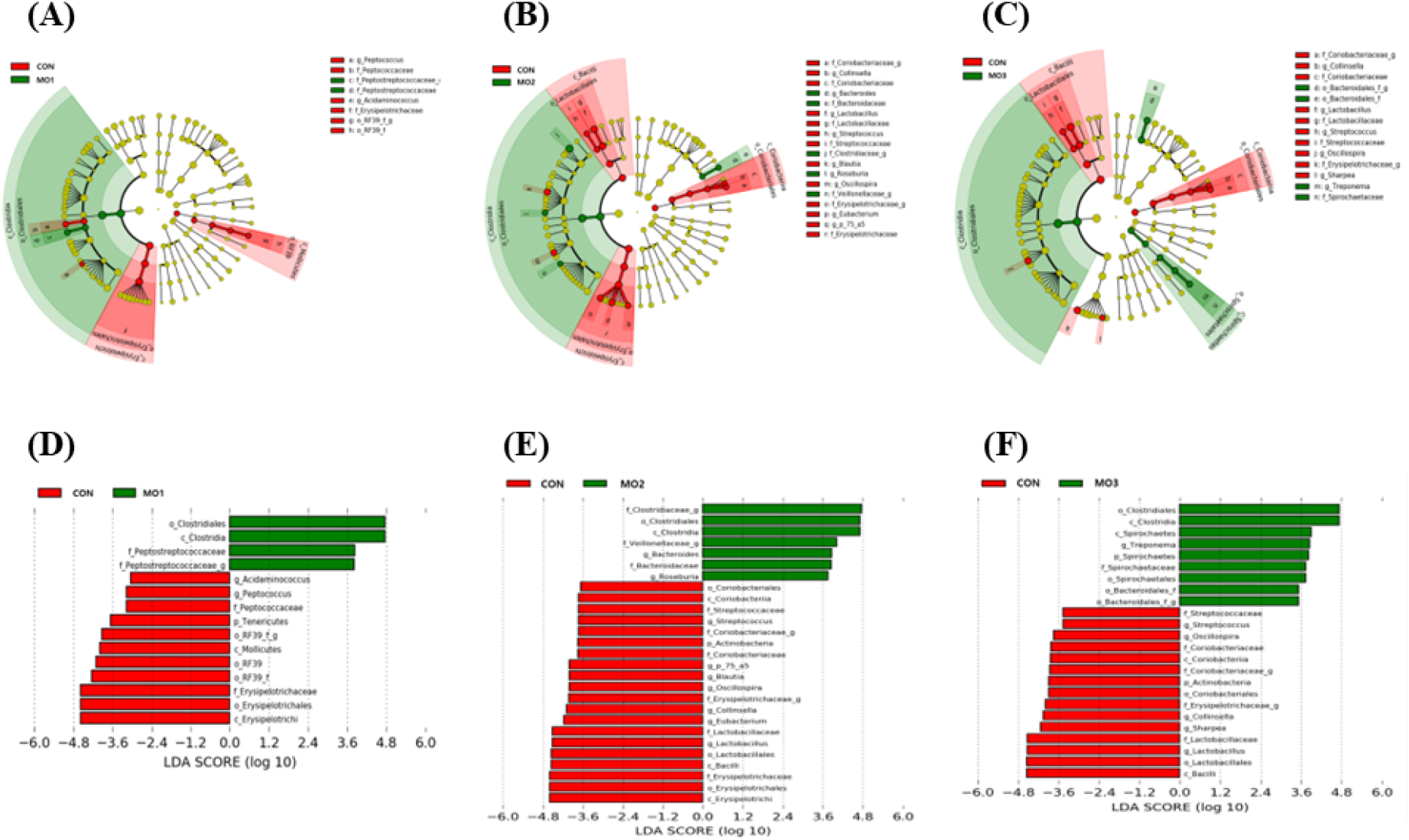
Fig. 4.
The distribution histogram of cladogram of bacterial taxa compared between CON and MO1 (A), CON and MO2 (B), CON and MO3 (C). Linear discriminant analysis (LDA) score between CON and MO1 (D), CON and MO2 (E), CON and MO3 (F). CON (control), no added; MO1, CON + monzogranite 0.1%; MO2, CON + monzogranite 0.2%; MO3, CON + monzogranite 0.3%.
Discussion
Interestingly, the different levels of dietary MON inclusion showed no effects on ADG, ADFI, and G/F, nevertheless, pathogenic microbiota was increased in MON MON-supplemented group. The MON possesses ion exchange, strong adsorption, and dispersion abilities due to its unique 2 : 1 layered structure (Hu et al., 2013). This structure enables MON to adsorb metabolically harmful agents like viruses, pathogens, ammonia, and numerous toxins in the intestine, subsequently through fecal excretion. Based on this mechanism, MON reduces damage to the mucous membrane system, maintains digestive tract health, and mitigates the occurrence of diarrhea in pigs (Song et al., 2012; Lee et al., 2020). Silica-based minerals have been evaluated to enhance the health and intestinal environment in weanling pigs over the past few years (Kong et al., 2004; Trckova et al., 2014; Liu et al., 2020; Zha et al., 2023). Similar to this study, silica-based mineral addition in a weanling pig diet did not show any prevention of post-weaning diarrhea and did not affect growth performance (Hu et al., 2013). Besides, the addition of 0.2% montmorillonite did not result in significant changes in the ADG, ADFI, and G/F of weaned piglets during the whole experimental period (Liu et al., 2020). These findings suggest that silica-based mineral alone has a limited effect on enhancing the growth performance of weaned piglets.
Despite alterations in gut microbiota composition, dietary supplementation with varying levels of MON did not influence the nutrient digestibility parameters measured, including DM, GE, CP, and EE. The observed increase in Clostridium abundance without corresponding enhancements in nutrient utilization implies that microbial shifts induced by MON may not have immediate functional benefits (Abbès et al., 2006). This suggests that while MON may affect the intestinal microbial environment, it does not necessarily translate into improvements in digestive efficiency over a 14-day feeding period.
In this study, the various levels of MON supplementation in weanling pigs over a 14-day period demonstrated that the decreased Chao1 index as a measure of α-diversity, along with changes in weighted, unweighted, and bray Curtis diversity as a measure of β-diversity. On the other hand, a lack of diversity can result in the proliferation of harmful bacteria, a condition known as dysbiosis (Mun et al., 2021; Lee et al., 2023). Dysbiosis of the gut microbiome can cause compositional changes that impact its functionality (Oh et al., 2021). In contrast, Liu et al. (2020) reported that the Shannon index, an α-diversity measure, was significantly higher in the ileum of the 0.2% montmorillonite supplementation in the weanling pigs as a silica-based mineral group compared to the control group. However, Zha et al. (2023) reported that the different levels of dietary attapulgite addition did not affect microbial α-diversity in weanling pigs. Increased bacterial diversity is indicative of a healthy and stable gut microbial community, which is beneficial to the host (Lee et al., 2023). These previous studies indicated that the silica-based mineral supplementation to the feed shows a high fluctuation in microbial diversity of weaned piglets regarding the source of silica-based minerals. The use of silica-based minerals in our study revealed the gut microbiota richness and diversity. Furthermore, the results of PCoA analyses demonstrated a significant difference in microbial community composition among the experimental groups, suggesting that MON alters the structure and composition of the gut microbiota in weaned piglets.
It is well-known that an imbalance of intestinal microbiota often occurs during the weaning stress in pigs, potentially allowing opportunistic pathogens to proliferate and causing gut-associated diseases (Konstantinov et al., 2004). Gut microbiota, which acts as microbial barriers within the intestinal mucosal barriers, are crucial for intestinal function and these functions include nutrient absorption, maintaining mucosal barrier homeostasis, immunomodulation, and defense against pathogens in pigs (Chen et al., 2018). In the present study, the MON supplementation treatment exhibited a greater relative abundance of the Clostridium group the Clostridium group contains the potential to cause diarrhea both before and after weaning which has the role as a primary pathogen that can cause pathogenic disease in piglets even though it is a component of the general gut microbiome in weaning pigs (Hosseindoust et al., 2017; Vasquez et al., 2022). The CON group exhibited a higher relative abundance of the Lactobacillus and Streptococcus groups. Lactobacillus, has compositive effects on anti-inflammation and antioxidants by enhancing gut microbiome balance (Jeong et al., 2019). Streptococcus group plays a significant role as a participant in nutrient utilization processes, including amino acid metabolism (Neis et al., 2015) and short-chain fatty acid production (Corr et al., 2009). Gastrointestinal health improvement is achieved by adding dietary silica-based minerals due to their greater absorption ability and toxin-binding role (Wang et al., 2021; An et al., 2023). In several previous studies, intestinal microbiota improvement was achieved by the addition of zinc-loaded silica-based minerals in pigs through the anti-microbial and pharmaceutical effects of Zinc (Li et al., 2021; Huang et al., 2023). On the other hand, the supplementation of attapulgite as a silica-based mineral did not dramatically affect the cecal microbiota environment (Zha et al., 2023). This inconsistency results from previous studies because different type of silica-based minerals used in the study has different absorptive abilities to combine with other agents.
Conclusion
Dietary MON inclusion decreased microbial richness and increased potential pathogenic bacteria, such as the Clostridium group. However, growth performance was not affected by adding different levels of MON. Silica-based minerals may adversely affect performance and alter the gut microbiome due to their different structure and processing method. Further research is necessary to elucidate how various sources of these minerals impact pigs differently.