Introduction
Fundamentals of Apoptosis
Apoptosis in Postmortem Muscle: Triggers and Evidence
Triggers of postmortem apoptosis: Energy depletion, oxidative damage, and calcium balance disruption
Evidence of postmortem apoptosis: Cell shrinkage and nuclear fragmentation
The Apoptotic Machinery and Its Regulation in Postmortem Muscle
Mitochondrial dysfunction and caspase cascade regulations
Caspase-independent pathways: The role of apoptosis-inducing factor
The role of lysosomes and signal amplification
Interaction with other cellular mechanisms
Inhibition mechanisms: Heat shock proteins and endogenous antioxidant system
How Meat Quality Changes by Apoptosis
Integrated Model and Implications
Conclusion
Introduction
Among meat quality attributes, tenderness is the most important factor in determining consumer satisfaction and repeated purchase (Miller, 2020). Tenderness is known to be determined by background toughness, degree of shortening, and aging potential. Among these, aging potential is a known key concept which is primarily determined by the enzymatic activities of calpain and calpastatin. However, the calpain-calpastatin model has limitations, as the activity of calpastatin accounts for only approximately 40 - 60% of the variation in tenderness (Shackelford et al., 1994). This gap prompted early recognition that other endogenous enzymatic systems must contribute to postmortem proteolysis (Sentandreu et al., 2002). Furthermore, the calpain model has a limitation to explain the continued tenderization that occurs long after µ-calpain (or calpain 1) activity has ceased, suggesting the involvement of complementary proteolytic systems (Ouali et al., 2006).
In this regard, it has been suggested that apoptosis acts as an early initiator of muscle structural changes (Ouali et al., 2006). The environmental shifts in postmortem muscle, such as profound hypoxia, severe energy depletion, and calcium overload, create conditions that are well-known to induce apoptosis (Sentandreu et al., 2002; Kemp and Parr, 2012). Cao et al. (2010) demonstrated the evidence of apoptosis, such as caspase activity increase, chromatin condensation, and DNA fragmentation occurred in early postmortem stage of beef. This is because the anoxic conditions caused by bleeding led to trigger apoptosis, mitochondrial dysfunction and cytoskeletal degradation (Rønning et al., 2017). These evidences of apoptosis in the postmortem muscle indicate that the muscle-to-meat conversion is driven by regulated cell death pathways leading to muscle structural disassembly.
This review presents a unified framework that emphasizes the central role of apoptosis in driving early muscle changes and its interplay with other proteolytic and protective systems. This integrated perspective offers a clearer explanation for muscle-specific tenderness differences and the diverse impacts of pre-slaughter stress, issues not fully addressed by previous studies.
Fundamentals of Apoptosis
Kerr et al. (1972) first described apoptosis as a “mechanism of cell deletion, which appears to play a complementary but opposite role to mitosis in the regulation of animal cell populations.” The events that occur within the apoptotic cell include shrinkage, plasma membrane blebbing, and ultimately the collapse of the cell into smaller fragments (Adrain and Martin, 2001). The function of the apoptotic process originally evolved in multicellular organisms as a means to eliminate slower-growing cells and prevent overgrowth (Vincent et al., 2013). Numerous factors may trigger apoptosis, including growth factor depletion, hypoxia, and DNA fragmentation (Lowe and Lin, 2000). Additionally, caspase-dependent apoptosis occurs via multiple signaling pathways that release cytochrome c (Cyt c) and activate caspase 3.
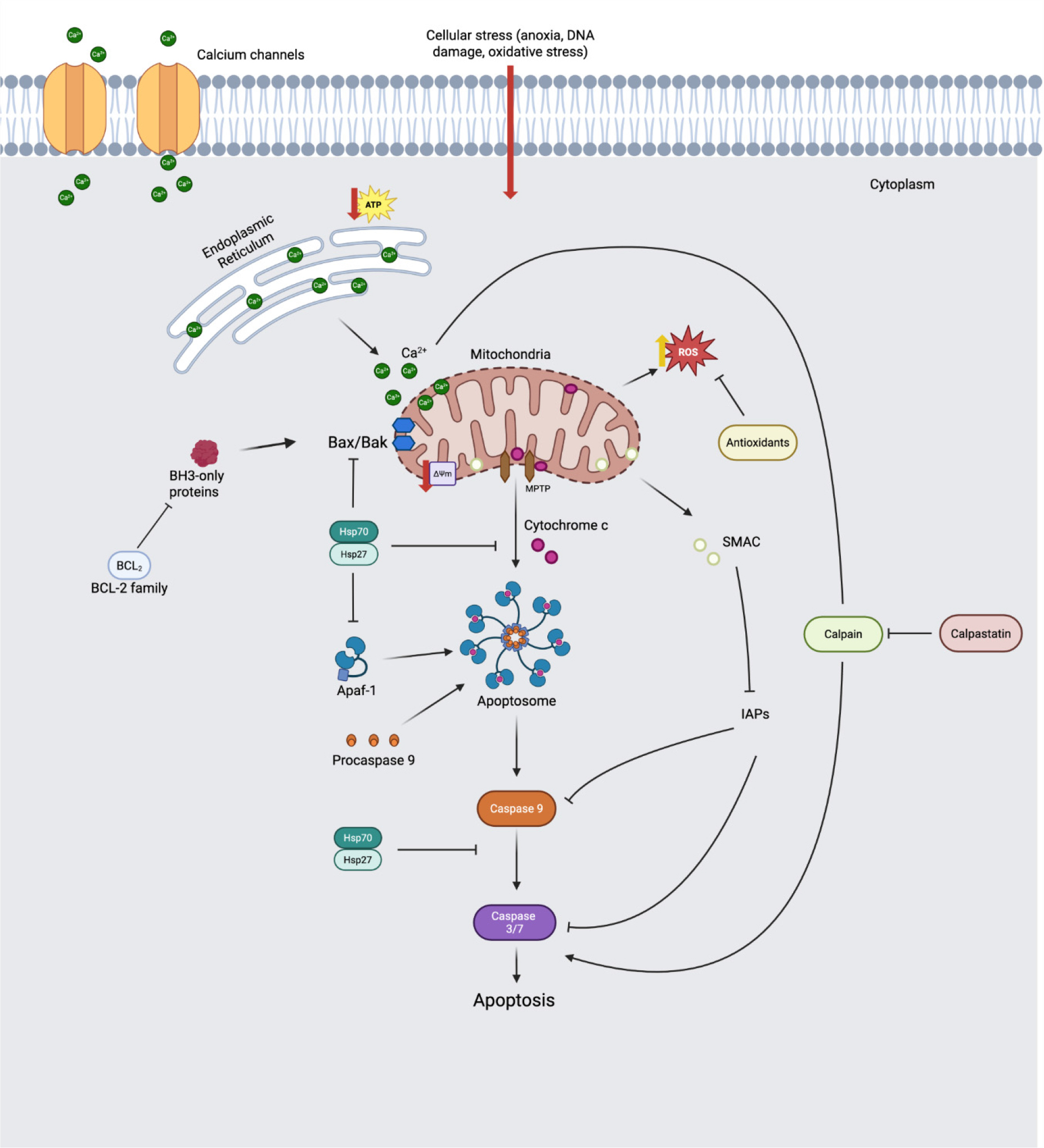
The execution of apoptosis is dependent on mitochondrial outer membrane permeabilization (MOMP), which is represented in Fig. 1. This process is regulated by the B-cell–lymphoma protein 2 (Bcl-2) family. Specifically, Bcl-2-associated X (Bax) is a pro-apoptotic protein that oligomerizes with Bax/Bcl-2 antagonist/killer (Bak) to induce MOMP, whereas Bcl-2 is an antiapoptotic protein that inhibits this formation (Lee and Choi, 2024; Ma et al., 2024b). These stress signals activate BH3-only proteins, such as Bid and Bim. These proteins function either by neutralizing anti-apoptotic proteins such as Bcl-2 and Bcl-xL, or by directly activating the effector pro-apoptotic proteins, Bax and Bak (Huang et al., 2016).
Cyt c is a protein that is 104 amino acid residues long and contains a heme group to help facilitate electron movement. It is located on the inner mitochondrial membrane (Green and Reed, 1998; Adrain and Martin, 2001; Ouali et al., 2006), and is the third protein to receive and pass on an electron in this procedure, passing it off to Cyt c oxidase in a final step that will convert oxygen molecules to water molecules (Hüttemann et al., 2011). About 15% of Cyt c is tightly bound to the inner mitochondrial membrane due to electrostatic and hydrostatic interactions, but the majority of Cyt c is loosely bound to membrane phospholipids (Cortese et al., 1995; Kagan et al., 2004). Cyt c also interacts with reactive oxygen species (ROS) to serve as an excellent antioxidant within the cell (Hüttemann et al., 2011). Solubilized Cyt c is often associated with inhibiting ROS formation, although tightly-bound Cyt c may be responsible for phospholipid peroxidation (Kagan et al., 2004). Cyt c is vital for apoptosis, as Li et al. (2000) found a marked reduction in caspase-3 activity in cells where Cyt c was knocked out.
Caspases are a family of cysteine peptidases that are aspartate-directed, in that their cleavage site is at a carboxyl-end Asp-XXX amino acid sequence. This function is suggested in the name provided by Alnemri et al. (1996), where ‘c’ indicates cysteine and ‘asp’ refers to the cleavage site at the aspartic acid residue. There are around 14 proteins belonging to the caspase family, and the structure of each protein consists of a large domain that, upon cleavage, becomes the active site for the caspase molecules and contains a cysteine and histidine residue, and a smaller domain which takes on a conformational role following cleavage (Fuentes-Prior and Salvesen, 2004). Effector caspases are activated from initiator caspases and are the enzymes most responsible for the lysis of structures within the cell, including cytoskeletal filaments (Adrain and Martin, 2001; Becila et al., 2010; Hüttemann et al., 2011).
In the intrinsic mitochondrial pathway, specific molecular steps involving Cyt c and caspase activation occur. In each pathway, a signal is relayed to activate the initiator caspase, which, in turn, activates effector caspases, allowing them to cleave cellular materials. Upon receiving an apoptotic stimulus, these binding interactions are disrupted, and Cyt c is released into the cytoplasm. In the cytoplasm, Cyt c must associate with Apoptotic protease-activating factor 1 (Apaf-1) and dATP/ATP to form a multimeric complex known as the apoptosome. The apoptosome serves as an essential activation platform for the initiator caspase, procaspase-9, thereby initiating the execution phase of apoptosis (Adrain and Martin, 2001).
Inhibitors of Apoptosis Proteins (IAPs) prevent apoptosis by directly binding to caspases or degrading them through ubiquitin ligase activity. For example, X-linked IAPs interfere with the dimerization of caspase-9 or directly inhibit caspase-3/7 (Namura, 2017). These anti-apoptosis pathways are controlled by Second Mitochondria-derived Activator of Caspases/Direct IAP Binding Protein with Low pI (Smac/DIABLO) is a pro-apoptotic protein located in the intermembrane space of mitochondria that neutralizes IAPs, thereby promoting apoptosis (Adrain et al., 2001).
Apoptosis in Postmortem Muscle: Triggers and Evidence
Triggers of postmortem apoptosis: Energy depletion, oxidative damage, and calcium balance disruption
The muscle-to-meat conversion process primarily involves energy depletion due to exsanguination. This rapid decrease in ATP and the resulting increase in the AMP/ATP ratio activate AMP-activated protein kinase (AMPK). AMPK plays a role in maintaining energy balance during the antemortem phase, but after death, it promotes irreversible apoptosis. Ma et al. (2022a) identified a significant increase (p < 0.01) in AMPK phosphorylation during the early postmortem period (0 - 24 h) in bovine longissimus dorsi, reaching maximum activity at 6 h compared to 0 h. This increase in the ratio was associated with approximately 25% mitochondrial dysfunction (e.g., loss of mitochondrial membrane potential) and a high correlation with caspase-3 activation. While this AMPK activation pathway is also observed in aging, the persistent AMPK activation induced by energy depletion after slaughter leads to cell death.
Changes in the postmortem environment, due to prior energy depletion and the development of hypoxia, cause electron leakage in the mitochondrial electron transport chain (Cadenas and Davies, 2000). This electron leakage in mitochondria, especially in Complex I and Complex III, occurs, and this electron transport causes the accumulation of ROS. These ROS, especially superoxide anions (O2•−) and peroxides, serve as key signals that trigger apoptosis. The production of these ROS induces oxidative stress, which is responsible for lipid peroxidation and the initiation of apoptosis signals (Green and Reed, 1998), and damages the bases and deoxyribose backbone of DNA molecules (Dizdaroglu et al., 2002). Wang et al. (2018b) found that when 20 mM H2O2 was applied to the longissimus lumborum region of the yak, this induced oxidative stress and augmented Cyt c release and led to a significant rise in Caspase-3 activity, peaking at 24 h (p < 0.05), confirming that ROS-mediated oxidative stress enhances the mitochondrial apoptotic pathway and yak meat tenderization.
This ROS-generated oxidative damage-induced apoptosis increases through specific upstream signaling pathways, most notably the Mitogen-Activated Protein Kinase-c-Jun N-terminal kinase (MAPK)-JNK pathway, which transmits the death signal to the mitochondria. ROS activates upstream kinases, leading to the sequential activation and phosphorylation of JNK. Activated JNK then translocates to the mitochondria, where it promotes apoptosis by inducing MOMP. This induced damage leads to Bcl-2 inhibition and Bax activation, disrupting mitochondrial membrane stability and promoting apoptosis. Ma et al. (2024a) found that when chicken breast was treated with H2O2, p-JNK-1 phosphorylation increased (p < 0.05), which in turn increased the downstream Bax/Bcl-2 ratio and caspase-3 activity.
These overall changes in ROS enhance the level of apoptosis initiation by oxidative damage to the lipids that compose the mitochondrial membrane (Cortese et al., 1995). Cardiolipins (CLs) in the inner mitochondrial membrane bind and immobilize Cyt c via electrostatic interactions. Cyt c exhibits a critical functional switch, acting as a cardiolipin-specific peroxidase in the presence of ROS (Kagan et al., 2005; Hüttemann et al., 2011). This conversion of Cyt c catalyzes mitochondrial membrane peroxidation, promoting its release into the cytosol (Kagan et al., 2005; Hüttemann et al., 2011). In addition to this leakage due to CL peroxidation, the sphingolipid messenger ceramide has also been shown to enhance membrane permeability. Zou et al. (2022) confirmed these lipid changes through mitochondrial lipidome analysis (LC-MS/MS) of bovine muscle (longissimus lumborum), finding that specific ceramide levels increased at 12 h postmortem. Notably, ceramide levels (Cer (d18:1/16:0)) increased (p < 0.05) at 12 h postmortem, consistent with the elevated ceramide content observed in LL muscle at 12 h postmortem.
In addition to AMPK activation, ROS production, and resulting lipid damage, disruption of calcium homeostasis is another factor that induces apoptosis. When the ATP-dependent Sarcoplasmic/Endoplasmic Reticulum Calcium ATPase (SERCA) pump is inactivated, calcium cannot be reabsorbed into the sarcoplasmic reticulum, resulting in a persistent increase in cytosolic Ca2+ concentration. This persistent increase in concentration triggers the unfolded protein response (UPR) and damages the endoplasmic reticulum (ER), which is responsible for calcium storage, resulting in cytotoxicity and subsequent meat quality defects. Guo et al. (2016) reported that Pale, Soft, Exudative (PSE) pork exhibited reduced SERCA1 expression compared to normal pork (p < 0.05), indicating compromised Ca2+ regulatory capacity. This correlated strongly with elevated ER stress markers, showing a 1.5-fold increase in GRP78 (Glucose-Regulated Protein 78) expression. Consequently, heightened apoptosis was observed in the PSE pork, evidenced by increased Caspase-3 activity and an elevated Bax/Bcl-2 ratio (p < 0.05).
Evidence of postmortem apoptosis: Cell shrinkage and nuclear fragmentation
Apoptosis induces a volume reduction of muscle cells, a regulated physiological process initiated by early apoptotic signals that modify ion channel activity. This change also causes water movement out of the cell due to changes in cell osmotic pressure. For example, Becila et al. (2010) utilized the hematoxylin-eosin staining method to confirm that the cross-sectional area of fibers in Wistar rat muscles decreased by approximately 30 - 33% over a 24 h postmortem period. In the same study, the concurrent loss of cell membrane asymmetry, which resulted in phosphatidylserine leakage, is also evidence of apoptosis.
Along with cell shrinkage, another sign of apoptosis is nuclear chromatin condensation (pyknosis) and subsequent nuclear fragmentation (karyorrhexis). This disruption of chromosomal structure is due to the activity of endogenous nucleases that specifically cleave DNA between nucleosomes. Cao et al. (2010) examined the nuclei of bovine muscle (longissimus dorsi) to visualize these nuclear changes. In this research, they detected condensation and localization of nuclear chromatin to the nuclear membrane on day 1 postmortem, followed by disintegration of apoptotic bodies on day 4. Furthermore, using the Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay, the positive nuclei increased (p < 0.05) postmortem, indicating an apoptotic response in postmortem muscle cells (Cao et al., 2010).
The Apoptotic Machinery and Its Regulation in Postmortem Muscle
Mitochondrial dysfunction and caspase cascade regulations
As previously described, post-mitochondrial stresses, such as oxidative stress and energy depletion, initiate apoptosis. These changes affect mitochondrial dynamics, the balance between fission (division) and fusion (Youle and Van Der Bliek, 2012). Postmortem mitochondrial stress causes proteins, such as Dynamin-related protein 1 (Drp1), to translocate to the mitochondrial surface, leading to mitochondrial fragmentation. This fragmentation destabilizes mitochondrial structure and promotes membrane reorganization, such as cristae remodeling. This remodeling promotes the efficiency of Bak oligomerization on the outer membrane, thereby increasing susceptibility to MOMP (Desagher and Martinou, 2000; Czabotar et al., 2014). Furthermore, Bcl-2 expression decreases over time postmortem (Zou et al., 2023), and the balance between Bak and Bcl-2 is disrupted, leading to increased MOMP during the postmortem process. This induces the leakage of substances such as Cyt c and Smac/DIABLO from mitochondria into the cytosol (Sierra and Oliván, 2013). Smac/DIABLO, released from mitochondria, neutralizes IAP and promotes caspase activation (Lv et al., 2019).
The caspase-dependent pathway is initiated following the release of Cyt c into the cytoplasm, where it binds to Apaf-1, inducing a conformational change that allows for the recruitment of procaspase-9 and the formation of the apoptosome (Adrain and Martin, 2001). The redox state of the heme iron of Cyt c determines this. The heme iron of Cyt c must be in an oxidized state (Fe3+) to adopt the specific conformational structure required for effective binding with Apaf-1; reduced Cyt c (Fe2+) has diminished affinity for Apaf-1. Wang et al. (2018a) confirmed that when the reducing agent TMPD (N,N,N′,N′-tetramethyl-p-phenylenediamine) (100 µM) was treated with yak meat extract, Cyt c was maintained in the Fe2+ state, which inhibited its binding to Apaf-1. As a result, caspase-3 activation was confirmed to be significantly suppressed by more than 50% at 12 h of postmortem compared with the control group. This suggests that the oxidative environment in the early postmortem period promotes apoptosome formation by oxidizing Cyt c (Wang et al., 2018a).
This caspase activity is sensitive to the rate of postmortem pH decline. Caspases generally exhibit optimal enzyme activity at pH 6.5 - 7.5. Acidification of postmortem muscles inhibits caspase activity. Ma et al. (2022b), however, confirmed that in Longissimus thoracis of crossbred bulls (Luxi yellow cattle × Simmental), the fast pH decline group (pH at 6 h < 5.8) showed higher caspase-3 activity at 12 - 168 h and lower shear force values (p < 0.05) than the slow pH decline group (pH at 6 h > 6.2). This suggests that a rapid pH decline promotes postmortem caspase-3 activation and apoptosis in bovine muscle, thereby accelerating tenderization. The contradictory phenomenon of high caspase-3 activity in the rapid pH drop group, despite caspase activity being optimal at neutral pH, might occur because the rapid pH drop triggers a stronger apoptotic signal via mitochondrial damage or lysosomal destabilization, leading to the release of Cyt c into the cytosol.
Caspase-independent pathways: The role of apoptosis-inducing factor
Apoptosis can also be induced by leakage of apoptosis-inducing factor (AIF), rather than by caspase alone. This pathway operates in parallel with the caspase pathway (Chen et al., 2020). When MOMP occurs, AIF is released from the intermembrane space and translocates to the nucleus (Norberg et al., 2010). Translocated AIF promotes chromatin condensation and DNA fragmentation through endonuclease activity, resulting in nucleolar disintegration (karyorrhexis). Zhang et al. (2018) observed a sustained increase in nuclear AIF accumulation in the longissimus dorsi of postmortem beef over a 7-day aging period, with a strong positive correlation (R2 = 0.798) between the nuclear accumulation and the myofibril fragmentation index (MFI). This suggests that caspase-independent pathways are activated during aging, potentially contributing to the deterioration of muscle structure.
The role of lysosomes and signal amplification
This apoptotic process is amplified by a positive feedback loop in lysosome-mitochondria interactions. ROS generated early after death oxidize lysosomal lipids, impairing their stability and leading to lysosomal membrane permeabilization (LMP). LMP causes iron (Fe2+) and lysosomal proteases (cathepsins) within the lysosome to leak into the cytoplasm. Leaked iron (Fe2+) catalyzes the Fenton reaction (Fe2+ + H2O2 → Fe3+ + ∙OH + OH−), producing highly reactive hydroxyl radicals (•OH), which dramatically escalate overall ROS levels and exacerbate oxidative damage (Held et al., 1996). Zhang et al. (2020b) demonstrated that treating postmortem beef (longissimus dorsi) with the iron chelator deferoxamine (DFO, 10 mM) reduced ROS production (p < 0.05) and subsequently inhibited Caspase-3 activity.
Concurrently, leaked proteases, such as Cathepsin D, cleave the cytosolic BH3-only protein Bid into its active, truncated form (tBid), which then translocates to the mitochondria to promote MOMP. Zhang et al. (2019) confirmed that increased Cathepsin D activity during aging of bovine longissimus dorsi correlated strongly (R = 0.903) with elevated tBid levels and enhanced caspase activation (p < 0.05), establishing a direct link between lysosomal leakage and the amplification of the mitochondrial apoptotic pathway. Therefore, iron ions and cathepsin release act as amplifiers that promote oxidative damage and MOMP, thereby driving continuous apoptosis.
Interaction with other cellular mechanisms
Autophagy, another degradation system that occurs concurrently with apoptosis, also exists. Autophagy, including mitophagy, delays apoptosis by removing damaged mitochondria, but when overactivated, it can contribute to autophagic cell death. Furthermore, recent studies have explored the correlation between autophagy and apoptosis, revealing key proteins such as Beclin-1 that are involved in regulating both pathways. Additionally, caspases can cleave autophagy-related proteins to modulate the process. For instance, Li et al. (2022a) investigated aging longissimus thoracis muscles of crossbred boars and reported that apoptosis was most intense at 12 h postmortem, followed by autophagy reaching its peak at 48 h postmortem. This type of autophagy or lysosomal pathway promotes the release of cathepsins, which can in turn amplify the mitochondrial pathway (Zhang et al., 2019). Furthermore, autophagy can be induced by inhibiting the PI3K/Akt/mTOR pathway, which in turn promotes softening by aiding in the degradation of myofibrillar proteins (Wang et al., 2022), and some studies attribute the differences in tenderization among varieties to autophagy activity (Garcia-Macia et al., 2014).
Additionally, the ubiquitin-proteasome system (UPS) is the primary non-lysosomal pathway for the targeted degradation of proteins. The UPS functions by tagging specific proteins with ubiquitin chains, marking them for degradation by the 26S proteasome complex. The UPS indirectly influences tenderization by regulating the degradation rates of key inhibitory proteins, such as calpastatin and heat shock proteins (HSPs). Liu et al. (2016) reported that when sheep longissimus lumborum muscles were treated with a proteasome inhibitor (MG-132), after 48 h, they observed a decrease in muscle ultrastructural destruction and a change in the pattern of myofibrillar protein degradation, suggesting a role for the UPS pathway in postmortem protein degradation. However, given the limited research, further investigation is necessary to fully support this hypothesis.
Inhibition mechanisms: Heat shock proteins and endogenous antioxidant system
The process of apoptosis is not only promoted in postmortem muscle, but there are also several mechanisms that inhibit this process (Fig. 1). HSPs are generally named based on their molecular weight. Both HSP 70 and 90 consist of an N-terminal ATPase domain and a C-terminal peptide-bonding region. Small heat shock proteins (sHSPs) are under 30 kDa in mass and are widely researched in terms of their roles in cancer and cell biology (Beere, 2005). A common structural feature of the sHSPs is the conserved alpha-crystallin domain (Lomiwes et al., 2014). HSPs can be activated and exhibit increased expression in response to various cellular or organismal stresses, including decreased pH, increased temperature, oxidative stress, or electrical shock (Beere, 2005; Lomiwes et al., 2014). These HSPs also function as a potent anti-apoptotic system, simultaneously inhibiting the apoptosis signaling pathway at multiple steps.
HSP70 and HSP90 have been shown to interfere with the interaction between Apaf-1 and procaspase 9, thereby preventing the progression of apoptosis. In addition, HSP27 can delay the detection of solubilized Cyt c induced by apoptotic factors, possibly by inducing Cyt c degradation upon its release from the mitochondria or by direct binding (Paul et al., 2002). These actions ultimately suppress downstream caspase activation (Beere, 2005). Additionally, HSP27 interacts directly with caspase-3, preventing its activation and subsequent proteolytic functions within the cell (Pandey et al., 2000; Voss et al., 2007). The practical impact of these inhibitory functions on meat quality is crucial. For instance, Hanwoo beef characterized by high αB-crystallin (a small HSP) expression at early postmortem (45 min) exhibited higher (p < 0.05) shear force (67.4 N vs 50.9 N) compared to the low expression group (Lee et al., 2022).
In addition to directly inhibiting components of the apoptotic pathway, a crucial function of HSPs is their capacity to chaperone other proteins. This involves facilitating correct protein folding and preventing the cleavage or aggregation of those proteins under stressful conditions (Lomiwes et al., 2014). Parsell and Lindquist (1993) emphasized that, during the process, HSP chaperones do not impart additional steric information but rather facilitate the protein’s self-coded folding protocol. HSP27 also regulates the polymerization of actin filaments, which is vital for muscle contraction and is a target for oxidative and proteolytic activity within the cell. They reported that αB-crystallin actively associates with µ-calpain in vitro, allowing µ-calpain to lyse αB-crystallin and thereby inhibiting calpain activity against myofibrillar proteins, such as desmin and titin (Lomiwes et al., 2014). Besides their protective function in folding, HSPs can prevent the degradation of myofilaments, including desmin, troponin T, and titin, in muscle cells under stressful conditions that may expose those regions of the sarcomere. For example, Kötter et al. (2014) demonstrated that sHSP binds to the titin interfilamentous region, specifically the N2B-us region, preventing the aggregation or degradation of myofilament proteins. Furthermore, activation of HSP25/27 (e.g., via oxidative stress-induced phosphorylation) specifically protects desmin from calpain proteolysis (Blunt et al., 2007).
Due to the protective functions of HSPs, the rate at which they are degraded and lose their function during postmortem aging is a key determinant of the rate and extent of tenderization. For instance, Ma and Kim (2020) reported that beef Longissimus lumborum (LL) muscle showed more extensive HSP27 degradation during postmortem storage compared to Psoas major (PM) muscle. This difference tended to coincide with reduced (p < 0.05) Warner-Bratzler shear force (WBSF) values and increased (p < 0.05) water-holding capacity (WHC) of postmortem in the LL muscle. In contrast, the PM muscle showed less HSP27 degradation and troponin-T degradation (p < 0.001), with limited improvement in tenderness (Ma and Kim, 2020). Furthermore, Cramer et al. (2018) consistently found lower levels of HSP27 degradation products in tough Callipyge lamb loins compared to normal lamb loins (p < 0.001), consistent with higher WBSF values (i.e., tougher meat) and reduced proteolysis (desmin, troponin T) in Callipyge meat. This evidence confirms that both the initial expression level and the degradation rate of HSPs are critical determinants of meat tenderness. Beyond the direct chaperone functions of HSPs, the cellular defense against apoptosis is also reinforced by the endogenous antioxidant system.
The endogenous antioxidant system serves as a critical defense mechanism that mitigates oxidative stress, thereby delaying the initiation and progression of apoptosis by neutralizing ROS. This system includes several key enzymatic defenses, such as Superoxide Dismutase (SOD), which exists in isoforms including cytosolic Cu, Zn-SOD and mitochondrial Mn-SOD (Landis and Tower, 2005). These catalyze superoxide radicals (O2•−) into hydrogen peroxide (H2O2) and oxygen (O2) via the reaction: 2O2•− + 2H+ → H2O2 + O2 (Valko et al., 2006). Subsequently, catalase (CAT) breaks down the generated H2O2 into water and oxygen: 2H2O2 → 2H2O + O2 (Fridovich, 1986). Glutathione peroxidase (GPx) eliminates organic peroxides (ROOH) by utilizing glutathione (GSH): 2GSH + ROOH → GSSG + ROH + H2O (Michiels et al., 1994). This finding aligns with Wang et al. (2023), who demonstrated that inhibiting Prdx6 activity in bovine muscle cells increased ROS levels, thereby enhancing Caspase-3/9 activity and accelerating desmin degradation. This suggests that the endogenous antioxidant system plays a role in delaying post-apoptotic events.
How Meat Quality Changes by Apoptosis
Apoptosis is induced during the muscle-to-meat conversion process, causing various biochemical reactions and influencing the meat quality characteristics perceived by consumers, as confirmed in Table 1. Apoptosis results in the activation of caspases, which degrade various proteins essential for the structural integrity of muscle fibers, such as desmin, titin, nebulin, and troponin-T (Chen et al., 2011). Actin is also a caspase target, cleaving it into a 31 kDa fragment. Longo et al. (2015) (Piedmontese cattle) and Laville et al. (2009) (Charolais cattle) found this specific marker in postmortem bovine muscle. These degradation products, as argued by Laville et al. (2009), correlate with meat tenderness. In their study, the ‘tender’ group of Charolais cattle longissimus thoracis muscle (average shear force approx. 27.7 N) exhibited a higher concentration of the 31 kDa actin fragment immediately post-slaughter (p < 0.05) compared to the ‘tough’ group (average shear force approx. 41.2 N), highlighting the functional relevance of apoptotic proteolysis in meat quality development. Recent advances in proteomic techniques, specifically degradomics (the identification of specific cleavage sites), have expanded the known repertoire of caspase substrates in muscle (Hanna et al., 2023). For example, He et al. (2025) confirmed that the degradation of vinculin and talin-2 was inhibited when a caspase-3 inhibitor was used to determine the effect of costamere protein on the tenderness of chicken breast, and immunofluorescence confirmed that caspase-3 was present at the same location as this protein at 3 and 24 h. Causal evidence provided by Zou et al. (2025) confirmed that inhibiting mitochondrial apoptosis with Mitoquinone (0.2 µM) reduced the MFI (p < 0.05) and delayed softening in beef.
Table 1.
Summary of studies on the effects of apoptosis on meat quality.
| Reference | Sample | Apoptosis detection | Key findings | Meat quality impact |
| Cao et al. (2010) |
Bovine, LD, PM, ST, N = 6 |
TEM, TUNEL assay, caspase-3 activity measurement |
Apoptosis actively occurs during aging, nuclear condensation at day 1 and apoptotic bodies present at day 4 |
Apoptosis contributes to meat tenderness development; Up to 6.5-fold change caspase-3 activity by 4 h postmortem. Apoptotic nuclei % at day 4: LD = 80%, PM = 66.7%, ST = 62.5% |
| Huang et al. (2016) |
Bovine, LD, N = 10 |
Western blot for Bax/Bcl-2 ratio, cyt c release, Caspase-9 and 3 activation |
Apoptosis via the intrinsic pathway is activated during beef aging, increased Bax/Bcl-2 ratio and cyt c release at day 3 |
Apoptosis begins 2 - 6 h postmortem, activity peaks at 12 h postmortem These findings suggest that these systems accelerate early postmortem proteolysis, leading to improved tenderness |
| Longo et al. (2015) |
Bovine, LD, N = 10 |
2D-PAGE and Western Blot |
Apoptosis is a core mechanism of meat quality change Detected apoptotic marker (31 kDa actin fragmentation) during aging |
Strong proteolysis of Troponin T and actin; Troponin T: 7.2-fold change at 44 days Actin: 2.5-fold change decrease at 10 days |
| Wang et al. (2018b) |
Bovine-Yak, LD, N = 10 |
Measure of ROS, cyt c release, Caspase-3 activity |
Oxidative stress induced by ROS promoted mitochondrial apoptosis and tenderization H2O2 treatment increased Caspase-3 activity 2.4 times |
ROS-mediated oxidative stress accelerated mitochondrial apoptosis, leading to earlier tenderization of yak meat; H2O2 treatment increased calcium 33.31%, Bax elevated to 1.86 ng/mL |
| Zhang et al. (2019) |
Bovine, LD, N = 6 |
Western Blot (cathepsin D activity, tBid levels, Caspase activity) |
Lysosomal leakage (cathepsin D) amplifies apoptosis by activating the mitochondrial pathway correlation between cathepsin D and tBid) |
Cathepsin D activity vs tBid level: R = 0.903 (p < 0.05) Associated with decreased WBSF. |
| Zhang et al. (2020b) |
Bovine, LD, N = 6 |
Iron chelator (DFO), ROS, caspase-3 |
Fe2+ promotes ROS generation amplifying apoptosis |
DFO treated vs Control: Decreased ROS production (p < 0.05), inhibited Caspase-3 activity, Increased WBSF |
| Zou et al. (2025) |
Bovine, LD, N = 6 |
Mitochondrial-targeted apoptosis inhibitor (Mitoquinone), Caspase-3 activity, MFI, TEM |
Mitochondrial apoptosis is the core mechanism inducing beef tenderization. Mitoquinone increased WBSF and inhibited Caspase-3 activity |
Inhibited MFI; 35.2 vs 58.6 (control) |
| Kim et al. (2023) |
Bovine, LT, N = 16 |
Grouping (HC/LC based on WBSF), Western Blot (cyt c, caspase-9/3, Desmin, Troponin T) |
Variation in tenderization is associated with apoptotic potential. The highly tenderized group (HC) showed significantly lower levels of apoptotic factors at day 14, correlated with higher rate of degradation of myofibrillar proteins |
Palatability variation (WBSF decrease); LC = 66.0 N vs HC = 52.4 N at day 14 |
| Chen et al. (2021) |
Broiler, Breast muscle, N = 4 |
Ultrasound treatment, Western Blot (Cyt c, Caspase-9/3), Cathepsin D/B activity |
Ultrasound promotes mitochondria and lysosome-mediated apoptosis. Increased cyt c release and activation of Caspase-9/3 and cathepsin D/B |
Enhanced tenderness (WBSF decrease). Ultrasound treated WBSF: 20.5 N vs control: 28.1 N |
| Wang et al. (2022) |
Broiler, Breast muscle, N = 8 |
Dietary Quercetin, Western Blot (autophagy/ apoptosis markers), Cathepsin activity |
Autophagy may contribute to tenderization by releasing lysosomal enzymes (Cathepsins) |
Enhanced tenderness. Shear force decreases as postmortem time extends. The quercetin treatment group was lower than the control (p < 0.01) |
| Yan et al. (2022) |
Broiler, Thigh muscle, N = 320 |
H202 treatment, RT-qPCR (Caspase-3, 6, 8, 9 mRNA), Western Blot (Caspase-3) |
Oxidative stress from H2O2 causes apoptosis and aberrant autophagy via the ROS/NF-kB pathway, reducing meat quality. Caspase mRNA and protein levels rose considerably. |
Impaired meat quality in 10% H2O2 Drip loss: 1.62% vs 1.25% (p < 0.01) Shear force: 20.09 N vs 15.36 N (p < 0.01) pH (24 h): 5.82 vs 6.07 (p < 0.01) |
| Zhang et al. (2020a) |
Porcine, LD, PM, N = 10 |
Western Blot (Caspase-3, Desmin degradation) |
Muscle type-specific effect; PM (oxidative) had higher Caspase-3 activity than LD (glycolytic) but less Desmin degradation |
PM vs LD: Higher Caspase-3 activity in PM, lower Desmin degradation. Higher WBSF. |
Apoptosis contributes to tenderization not only through direct proteolysis but also by synergizing with the calpain system, primarily by degrading calpastatin, the endogenous calpain inhibitor (Pörn-Ares et al., 1998). Calpastatin is a large protein composed of four repeating and homologous inhibitory domains, approximately 140 amino acids each (Maki et al., 1987). One molecule of calpastatin inhibits multiple calpain molecules, as each domain may bind to a calpain (Wendt et al., 2004). It functions as a competitive inhibitor by binding directly to the active site of µ-calpain via its central consensus sequence (Croall and McGrody, 1994). Cleaving calpastatin suppresses the competitive inhibitory effect, thereby enhancing the effectiveness of the calpain system. This synergistic effect has been confirmed in the Callipyge sheep model. These sheep are known to have high calpastatin activity (Delgado et al., 2001), but subsequent studies have shown that they exhibit lower (p < 0.05) caspase-3/7 activity (Kemp et al., 2009). This reduced caspase activity prevents a larger-than-normal amount of calpastatin from being sufficiently degraded, resulting in persistent inhibition of calpain activity and the production of tougher meat.
However, the direct contribution of caspases to tenderization has been a topic of debate. Kemp et al. (2006) provided early evidence for this relationship, finding that higher activities of Caspase-3/7 and Caspase-9 early postmortem (0 - 32 h) in porcine longissimus muscle were significantly associated with lower shear force at 192 h (r = -0.62 and r = -0.68, respectively). Underwood et al. (2008) reported that caspase-3 activation was not detected in postmortem beef and was not correlated with shear force, suggesting that it is unlikely to be involved in tenderization. This discrepancy might be attributed to methodological factors, such as limitations of ex vivo muscle strip models, redundancy in proteolytic systems, or suboptimal conditions for inhibitor efficacy. Despite this controversy, accumulating evidence using more sensitive detection methods, specific inhibitors, and causal studies strongly supports the regulatory and proteolytic roles of caspases in the tenderization process across various species (Laville et al., 2009; Chen et al., 2011; Li et al., 2022b; Zou et al., 2025).
While most studies have examined the effect of apoptosis on tenderness, recent research suggests that it is also related to other meat quality traits, particularly WHC, color, and oxidative stability. As previously described, apoptosis causes cell shrinkage, resulting in the formation of physical drip channels between muscle fiber bundles. These drip channels allow water to leak out due to the force of gravity. Indeed, Yang et al. (2021) analyzed pork using transmission electron microscopy (TEM) and confirmed that drip channel formation accounts for weight loss and is associated with desmin degradation. Furthermore, as the connection between myofibrils and the cell membrane weakens, the ability to retain water is lost. This mechanism is currently being investigated in some studies. For example, Zhang et al. (2013) observed a significant correlation between the number of apoptotic cells and cooking loss in breast and thigh meat of ducks (breast: r = 0.556; thigh: r = 0.848). Although not a livestock species, a very high correlation (r = 0.982) between drip loss and apoptotic nuclei was also observed in fish (Tie et al., 2022).
As with WHC, the correlation between meat color and oxidative stability, as well as apoptosis, has recently been confirmed. Tuell et al. (2021) confirmed that when arginine supplementation delayed apoptosis by producing Nitric Oxide (NO), meat color stability in beef was improved over a 28-day aging period. This suggests that apoptosis causes mitochondrial dysfunction, leading to reduced mitochondrial oxygen consumption and metmyoglobin-reducing activity, both of which are associated with meat color stability. This loss of function accelerates myoglobin oxidation and reduces meat color stability. Zhang et al. (2013) also confirmed that there was a correlation between apoptosis and L* (breast:r = 0.800; thigh: r = 0.781), a* (breast: r = 0.847; thigh: r = 0.851), and b* (breast: r = 0.878; thigh: r = 0.568) values in duck.
Integrated Model and Implications
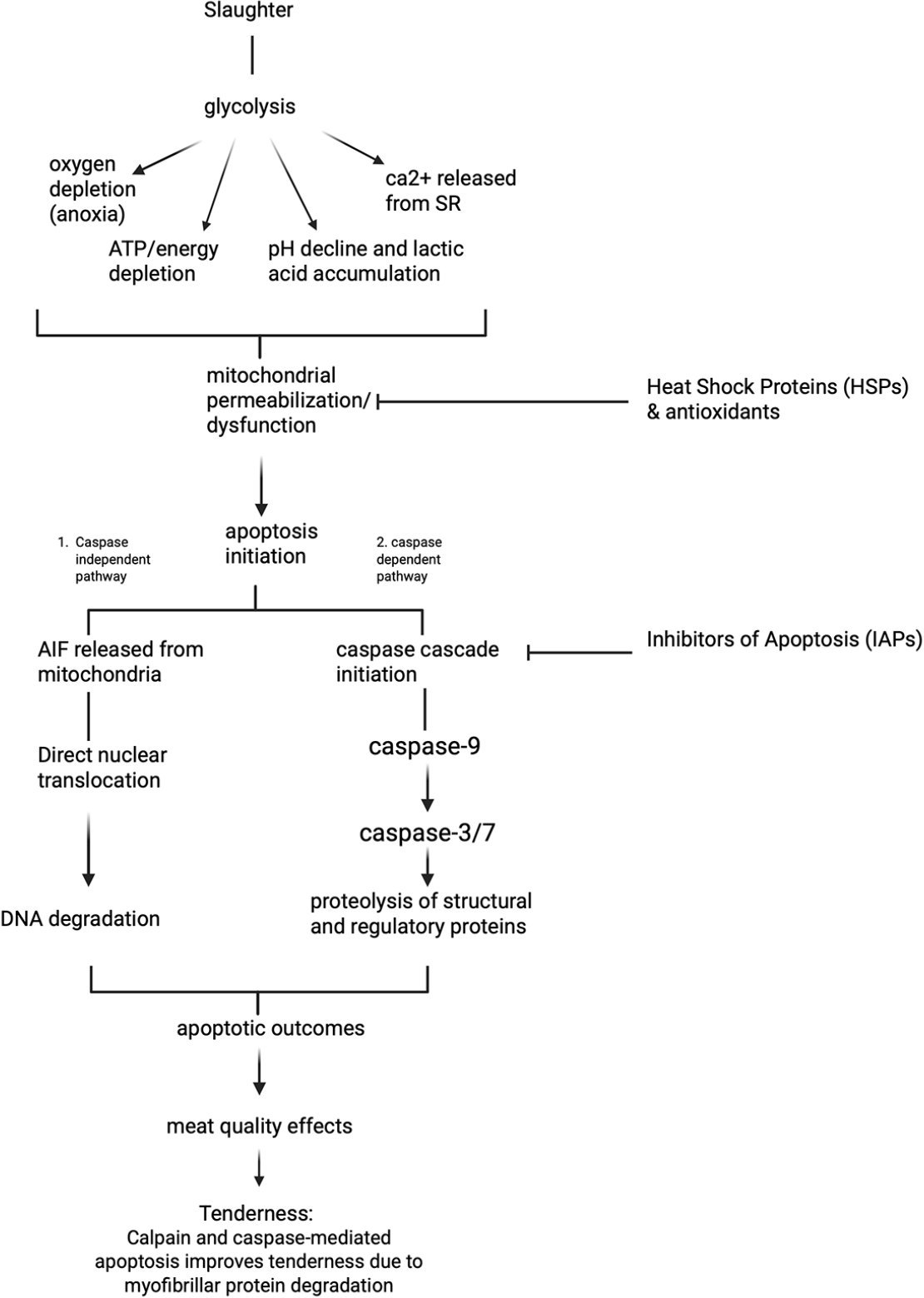
This review demonstrates that the muscle-to-meat conversion process is not simply a consequence of changes driven by the traditional calpain system, but rather a consequence of interactions triggered by the initiation of apoptosis. Fig. 2 represents the overview of apoptosis pathways related to meat quality. When postmortem environmental changes initiate apoptosis, it participates in tenderization through direct caspase degradation and the stimulation of the calpain system. These caspase changes accelerate the rate of tenderization through a feedback loop. Calpain and caspase mediated apoptosis improves meat tenderness due to myofibrillar protein degradation. Although limited research exists and further support is needed, it is suggested that apoptosis may induce cytoskeletal degradation. This structural change disrupts the linkage between the myofibrils and the sarcolemma, promotes drip channel formation. Myowater leaks into the extramyofibrillar space, finally increasing drip loss. Furthermore, mitochondrial dysfunction induced by apoptosis may lead to the loss of oxygen consumption ability of mitochondria and accelerate metmyoglobin formation. This apoptotic engine is inhibited by HSPs and endogenous antioxidant enzymes, which function as antagonists.
However, the relationship between apoptosis and meat tenderization may vary across muscle sites. This is due to differences in muscle type: oxidative or glycolytic. Zhang et al. (2020a) found that PM had more vigorous caspase-3 activity than longissimus dorsi, but actual protein degradation (desmin) was lower. This is because oxidative muscles inherently maintain higher baseline expression levels of HSPs (e.g., αβ-crystallin) and greater antioxidant capacity to counteract oxidative stress (Lee et al., 2022). Ma and Kim (2020) reported that HSP27 degradation was slower in bovine PM than in LL. Moreover, Zhang et al. (2022) reported that differences in the protein’s phosphorylation status were also a factor. In other words, even if a strong apoptotic signal is observed, the actual degradation capacity can differ depending on the strength of the protective system.
The impact of apoptosis on meat quality is highly context-dependent, often yielding opposing outcomes depending on whether the stress-inducing factor occurs during the antemortem or postmortem stage. For instance, while postmortem treatment with H2O2 enhanced tenderness (Wang et al., 2018b), similar oxidative stress induced antemortem resulted in decreased tenderness (shear force increased from 15.36 N to 20.09 N) and increased drip loss (1.25% to 1.62%) in broiler thigh muscle (Yan et al., 2022). This difference is explained by the differential cellular responses based on the timing of the stress. Ante-mortem stress (e.g., heat stress, exercise) disrupts the homeostasis of ‘living’ cells, triggering a robust adaptive defense mechanism to ensure survival. This defense involves the rapid upregulation of cytoprotective proteins, such as HSPs (Salo et al., 1991; Blunt et al., 2007), and the enhancement of antioxidant enzyme activities. For example, broiler chickens subjected to acute heat stress showed increased CAT, SOD, and GPx activity levels as a coping mechanism (Altan et al., 2003; Azad et al., 2010). This upregulated defense system persists in the early postmortem period (carryover effect), inhibiting protease activity and therefore resulting in tougher meat. In contrast, postmortem interventions apply stress to cells incapable of mounting a defense response, thereby unilaterally promoting apoptotic pathways and accelerating protein degradation in the absence of resistance.
This link is consistently observed in industrial conditions. Malheiros et al. (2021) confirmed that the very tough meat group of the Nellore breed exhibited higher HSP expression levels than the tender group. Furthermore, older goats with tough meat showed higher mRNA and protein expression levels of HSPs and sHSPs than young goats (Saccà et al., 2019). This is a practical example of how antemortem heat stress induces increased HSP expression and, in turn, decreases tenderness by inhibiting postmortem proteolytic enzymes.
Understanding the mechanisms of muscle-to-meat conversion via apoptosis offers strategic opportunities to improve meat quality within the industry. Electrical Stimulation (ES), a widely used post-slaughter technology, enhances tenderness, but its effect on apoptosis is complex. ES accelerates ATP depletion and causes massive Ca2+ release, which can increase mitochondrial stress and promote the initiation of apoptosis. Chen et al. (2025) found that application of ES significantly accelerated ATP and glycogen depletion, leading to a rapid decrease in pH. This process dramatically increased the activities of Ca2+-ATPase and Na+-K+-ATPase, which directly triggered the mitochondrial Ca2+ overload and the mitochondrial apoptosis cascade. This was demonstrated by an increase in the apoptosis rate at 24 h, enhanced caspase-9 and -3 activities, and, consequently the degradation of key structural proteins such as desmin and troponin-T. This study clearly demonstrates that ES can precisely regulate apoptosis, significantly reducing shear force and improving quality of the final product. However, the overall effect requires optimization, as the rapid pH drop induced by ES can simultaneously inhibit caspase activity (Ma et al., 2022b), illustrating the need for precise control to balance accelerated metabolism with optimal proteolytic conditions. Emerging technologies like ultrasound can enhance tenderness by altering membrane permeability and inducing ROS generation. Chen et al. (2021) confirmed that ultrasound treatment (40 kHz, 150 W, 30 min) on chicken meat reduced shear force (20.5 N vs 28.1 N, p < 0.05) by promoting both mitochondrial and lysosomal-mediated apoptosis.
Conclusion
The meat tenderization process is not solely driven by the traditional calpain-mediated protein degradation system but is a complex, dynamic biochemical process orchestrated by apoptosis induced by postmortem environmental changes. Hypoxia, energy depletion, oxidative stress, and calcium overload, which occur immediately after slaughter, activate the mitochondrial-mediated apoptotic pathway, triggering the activation of the caspase cascade. The integrative model proposed in this review demonstrates that activated caspases not only directly degrade muscle structural proteins but also promote the calpain system by degrading calpastatin, a calpain inhibitor, thereby enhancing tenderness. However, this process may have adverse impacts on WHC, color, and oxidative stability. This apoptotic process is strongly inhibited by HSPs and the endogenous antioxidant system. HSPs may play a central role in determining the rate and extent of tenderness development by blocking apoptotic signals and acting as chaperones to protect proteins. This integrative model provides important insights into several challenging issues in meat science. First, differences in muscle fiber type (oxidative vs. glycolytic) can be explained by differences in HSP expression levels and antioxidant capacity in each muscle. Second, antemortem stress degrades meat quality because living cells increase HSP expression as a defense mechanism, which persists until after death and suppresses proteolytic enzyme activity. In contrast, postmortem stress promotes apoptosis without a defense response. In conclusion, meat quality is determined by the balance between the apoptotic and proteolytic systems and the HSP and antioxidant systems. Understanding this integrated mechanism will provide a strategic foundation for optimizing postmortem treatment techniques, such as electrical stimulation, and ultrasound and enhancing the efficiency of high-quality meat production.